Do you aspire to design an innovative medical device? But how can you be sure you’re ready to take the plunge? How can you chart a course towards certification for your medical device, and thus towards the success of your
Do you aspire to design an innovative medical device? But how can you be sure you’re ready to take the plunge? How can you chart a course towards certification for your medical device, and thus towards the success of your

Do you aspire to design an innovative medical device?
But how can you be sure you’re ready to take the plunge?
How can you chart a course towards certification for your medical device, and thus towards the success of your project?
You need to stay on track, despite the rollercoaster ride, until you reach your destination.
Designing a medical device is ambitious, and above all, it is to your credit. A medical device is a healthcare product intended for use in the diagnosis, prevention, monitoring, treatment or alleviation of a disease or injury.
There are different types of medical devices. These devices do not pose the same level of risk to consumers. If the product you wish to develop meets the characteristics of a medical device, it will be essential to define its class.
Medical devices are classified into four categories: I, IIa, IIb and III. Classification depends on the level of risk posed by the device and is defined in accordance with Article 51 and Annex VIII of the European Medical Devices Regulation 2017/745.
All these innovations are essential to the development of our current healthcare system and contribute to improving patient care and optimising the organisation of healthcare.
How do you create a medical device?
Consider the common thread running through your project: innovation! You need to come up with a product that is useful, of course, but your goal is to make it indispensable.
Let’s find out together what steps are needed to design this medical device!👇
Launching an innovative medical device project can be an exciting journey, but one that is sometimes fraught with pitfalls. That is why it is important to frame it correctly from the outset.
The first challenge is to create a new promise through your product.
You must take into account all the factors that will influence your project at one point or another.
A crucial aspect of medical device development is the regulatory and standards environment.
You must ask yourself the right questions in order to secure your project. This basically involves a number of steps:
Scientific monitoring will be necessary and will be of great help in supporting you throughout the project. This step will help you determine whether your product will meet market needs and whether there is sufficient demand for your product.
It is important to work with people who have experience in quality and regulatory affairs to ensure full compliance with all requirements. There are consultants in this field, such as MD101. Their support can also be of great help in compiling all your documentation.
Developing a development strategy allows you to define the quality standards to be met, regulatory requirements, development deadlines and associated costs.
You are probably already aware of everything you need, but it is important to highlight these key steps that will support you in launching your project.
As you are about to invest time, money and numerous resources in this project, you need to secure it.
Properly framing your project and identifying the necessary resources is therefore the key to a good start. 🔑
In France, the market is controlled. When a medical device does not comply with legislation or regulations, or poses a health risk, it may be withdrawn or even recalled from the market, which may result in a temporary or permanent suspension of its marketing authorisation.
In Europe, the development of medical devices is governed by the new MDR 2017/745 regulation, which is supplemented by several standards (in the case of electronics, software or the use of substances).
At the end of the development process, if the product is deemed to comply with the regulation and applicable standards/requirements, it will be awarded the CE mark.
The European medical device market is therefore subject to very strict regulations that you will be required to comply with in order to obtain certification for your medical device and, ultimately, to be able to launch it on the market.
This is why a thorough understanding of the market is necessary. As a project leader, you may not have the necessary resources. Calling on an expert can be a great help.
The evolution of current standards clearly illustrates this need.
We are seeing radical changes with companies that are sometimes quite young and do not always have the resources or expertise in standards and regulations that people necessarily have, and sometimes need to review their entire strategy to meet the new requirements.
All of this can lead to significant additional costs and/or extra work. This is where experts will play an important role and add significant value.
A team of experts handles a large number of topics each year, giving them a broad overview. The team also has knowledge and expertise in the requirements of notified bodies.
All of this provides insight into:
The involvement of an expert allows for a targeted response, tailored to the client’s needs and facilitating the transition to the organisation. This support is provided on a case-by-case basis, with different strategies and varying priorities.
Surrounding yourself with a qualified team allows you to draw on skills and resources that you may not always have as a project leader. For example, if a device requires expertise in mechanics, electronics or other areas, you can find these skills among experts.
Today, the guiding principle of a medical device project is compliance. To successfully produce your device, you need a quality system that encompasses your entire project.
In particular, you will need to draft documentation covering your entire project in order to submit it to a standards body.
How do you know which documents to provide, what evidence to submit, and which steps to include? 🤔
These issues must be considered at the outset of the project, as failing to take everything into account from the start can pose a risk. You could end up with significant additional costs and a much longer time to market.
Many experts assist their clients in implementing their overall strategy, including testing, verification, protocol drafting and product monitoring.
It is these skills and the expertise of these specialists that will help you secure your project. Whether or not you have internal resources, an expert’s perspective will always add value.
You now have all the information you need to carry out your project.
It is essential to prove the value of your concept in order to convince potential future industrial partners as well as the certification body. You will therefore need to prove that your solution:
Now you can begin demonstrating technical feasibility.
This will determine whether your product can be developed with the resources you have available and whether the technologies needed for its development already exist or need to be created.
To do this, you need to carry out a Proof of Concept (PoC).
This PoC is the first stage of development. This ‘classic’ prototype is a proof that does not need to be tested in real conditions; it simply needs to demonstrate feasibility.
As long as your medical device is in PoC form, you have no documentation requirements, the tests you carry out do not provide any insight into the finished product, and you therefore have a certain amount of freedom.
But development does not stop there.
You must make your prototype compatible with the use you are seeking to demonstrate.
If you are creating a medical device that is intended to be worn, use materials that are suitable for skin contact and stable. If you define your final materials as soon as you create the prototype, you will be able to anticipate the production of your product.
Once your PoC is ready and validated, you will be able to test your concept in real life and prove its benefits in use.
As you can see, what differentiates the development process for a medical device from that of other innovative products is the mandatory testing and trial phases that are specific to medical devices.
All the data collected during these testing phases will be used to document your project and have it validated by an approved body so that it can ultimately be marketed.
Testing a product under development is common to all types of products.
However, this stage is particularly important in the context of a medical device, as it will assess the safety and effectiveness of the device under development. It also involves a number of tasks, such as the formalisation of specific files, which will be necessary for the validation of the final medical device.
There is a clear common thread in the development of a medical device: compliance. The development of the device requires a comprehensive quality system that encompasses the entire project.
In particular, preclinical and clinical studies must be conducted to prove the benefits, and all documentary evidence must be recorded in order to submit the file to the body that grants marketing authorisation.
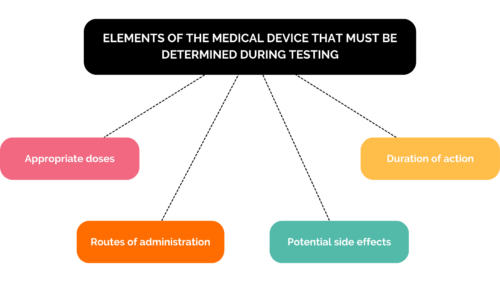
You will therefore need to develop testing, verification and validation strategies, as well as implementing protocol drafting, test automation and product monitoring on the market.
There are two types of development phase in the medical device sector:
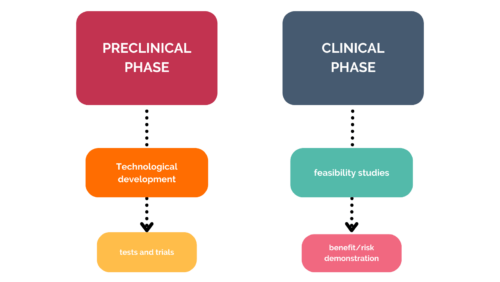
Preclinical trials will not provide the same answers as clinical trials. It is a development process that aims to make the prototype as reliable as possible, so that clinical trials can then be carried out.
It is during these phases that you will demonstrate the value of your new product. Throughout the development of this medical device and before conducting your clinical trials, you will need to consider:
Clinical trials, on the other hand, are tests carried out under near-real conditions. The device is tested in a restricted setting, but the aim is still to demonstrate the benefits it can and should bring. To do this, the trials are carried out directly on the ‘target’.
This scientific study aims to assess the non-toxicity, efficacy and human tolerance of a new diagnostic or therapeutic method or treatment.
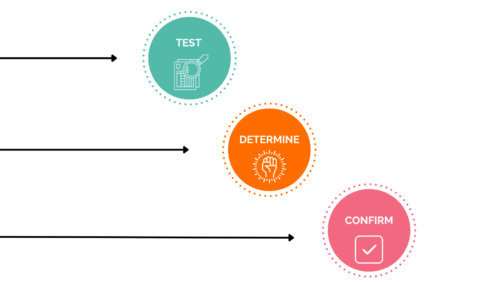
In conclusion, these trials allow you to test your product, determine the benefits it can offer and finally confirm the value of your innovation.
Keep in mind that conducting these trials is subject to regulations that provide a protective framework for all potential users. These regulations guarantee safety. That is why good support can be a considerable help, for example when drafting documentation.
What is the purpose of these trials?
As you will have understood, clinical trials must be integrated into your medical device development process.
There are then three final steps in the testing process:
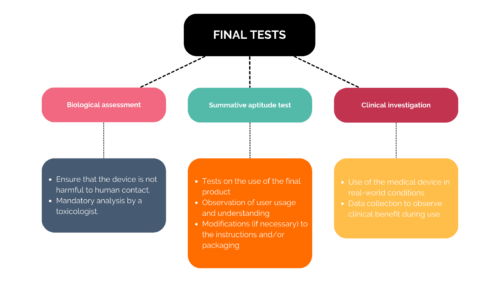
The results of all your trials will be reviewed by regulatory authorities before the medical device is approved. They are subject to significant regulations, which is why expert guidance can be beneficial.
This is known as regulatory approval: it is a necessary step in bringing a medical device to market.
Regulators require solid evidence of a product’s efficacy and safety before approving it for use on the market.
The data collected during testing is therefore used to meet regulatory requirements and obtain approval from regulatory authorities.
You are approaching the final hurdle!
As we have been saying since the beginning of this article, the ultimate goal is to get your product to market. It is the regulatory approval of your medical device that will allow you to move on to the final stage: marketing.
This is a difficult stage that must be anticipated. The approval of your medical device and its classification is certainly not within your remit, but it is up to you to do everything possible to ensure that this stage goes smoothly.
But how can we talk about challenges when it is an organisation that is responsible for approving or rejecting the device for market launch?
Quite simply because, during your long development process, you will normally have compiled a complete documentation file proving the value of your innovation and the benefits it brings.
An incomplete file can have serious consequences, such as:
It could be said that certification only comes at the end of the project, but it is like the industrialisation of your product: it needs to be defined as early as possible and prepared throughout your project.
As you can see, developing an innovative medical device is a unique process.
But you’re here now, you’ve stayed on track despite everything. 💪
By following the key steps with project scoping, proving the value of your concept, and conducting all the rigorous pre-clinical and clinical tests and trials to obtain certification, project leaders can significantly improve the chances of success for their medical device on the market.
To find out more about our support for project leaders specialising in the medical device sector, contact us!
Un peu de lecture
Des articles, des podcasts, des webinars… et surtout des conseils pratiques ! En bref, une collection de ressources pour mener à bien votre projet.